|
|
Kezdőlap-Home
Page |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
II. évfolyam 3. szám 2001. július [HUN] - Magyar cikk
|
On the Possibility to Produce a MgB2 Superconductor Layer by Electrochemical Synthesis from Molten Salts G.Kaptay LIMOS R&D, Department of Physical Chemistry, Faculty of Materials and Metallurgical Engineering, University of Miskolc, 3515 Hungary, Miskolc, Egyetemvaros
On January 10, 2001, Jun Akimitsu announced his discovery at the Symposium on Transition Metal Oxides in Sendai, Japan, on the 40 K superconductivity of MgB2 [1]. Two months later, at the meeting of the American Physical Society 78 papers were presented, stimulated by Akimitsu’s discovery [2]. The research group of the author has been working for the last decade on the synthesis of borides from molten salts, mainly that of refractory metals (Ti, etc.). As follows from our recent review [3], in the more than 100 papers devoted to the subject of electrochemical synthesis of borides from molten salts none of them dealt with the synthesis of MgB2. The reason is that this boride was not considered to be ‘interesting’ due to its mechanical properties. After the discovery of its high-temperature superconductivity, in 2001-2002 more papers are expected to be published on MgB2, than total before 2001. The aim of this short paper is to study the thermodynamic possibility of electrochemical synthesis of the stoichiometric MgB2 layer from a molten salt. This analysis will be performed by constructing the so-called EES Diagram (Equilibrium Electrochemical Synthesis Diagram) for the Mg-B system. The principles of constructing binary and ternary EES diagrams have been developed by us earlier [4-6].
There is now measured phase diagram is known for the Mg-B system. According to the review of Samsonov et al [7], there are four stable borides in the Mg-B system: MgB2, MgB4, MgB6 and MgB12. The thermodynamic properties of theses phases are given in Table 1, according to [7, 8]. One can see the large difference in heat of formation data given in [7, 8]. In the recent review of Predel [9] the tentative phase diagram for the Mg-B system is published, based on data of Spear [10], with only three compounds (MgB2, MgB4 and MgB6), showing much higher decomposition temperatures compared to [8] (see Table 1), but with no thermodynamic data et al, despite the title of the handbook [9]. In the present work, thermodynamic data from the later, and probably more reliable source of [8] will be accepted. This choice also means that our calculations are limited to Mg-B diagram with the B/Mg mole ratio £ 4. However, as our aim is to study the possibility to deposit MgB2, this limitation is acceptable for us. It should also be noted, that calculations are limited below 1,100 K by the decomposition temperature of MgB4[8]. At higher temperature, the properties of MgB6 are needed to calculate the conditions of MgB2 electrodeposition. This limitation is not catastrophic from the practical point of view either, as the common basic electrolyte for electrochemical synthesis of borides is the NaCl-KCl eutectic, allowing to perform the synthesis around 700 oC (1,073 K). The standard Gibbs energies of formation of the two magnesium borides between 700 and 1,100 K are given in Table 2 [8] (with pure Mg and B as reference state). Table 1. Thermodynamic properties of magnesium borides
*: decomposition temperature Table 2. Standard Gibbs energies of formation (kJ/mol) of MgB2 and MgB4 [8]
3. Calculation of the EES diagram for the Mg-B system As has been shown by us previously, the equilibrium synthesis-potential of a compound MgBq from a molten salt, containing Mg+2 and B+3 ions on an inert cathode can be calculated based on (electro-)chemical thermodynamics as [4-6]:
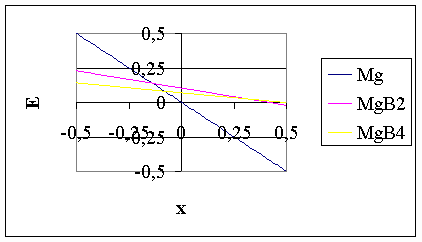
where q – is the stoichiometric coefficient of the MgBq compound (q = 2 or 4), F – the Faraday constant, x = EBinert – EMginert, i.e. the difference between the equilibrium depositions potentials of B and Mg on an inert cathode, without compound formation. As common in electrochemical thermodynamics, the choice of reference electrode doesn’t have any influence on the final conclusions. Therefore, in Eq.(1), B+3/B is considered as a reference electrode, i.e. the deposition potential of boron is taken as zero: EBinert = 0, and EMginert = -x. Hence, in the given system four different cathodic products can be deposited at low current density (near equilibrium): B, with EBinert = 0, Mg with EMginert = -x, and the two borides, MgB2 and MgB4, with a synthesis potential determined by Eq.(1). At different values of parameter x, the cathodic product will be also different. The equilibrium cathodic product will be that, having the most positive deposition (synthesis) potential. The deposition (synthesis) potentials are shown as function of parameter x in Fig 1 for T = 1,000 K.
Fig.1. Equilibrium deposition and synthesis potentials of possible cathodic products from a molten salt consisting of Mg+2 and B+3 ions at 1,000 K One can see from Fig.1, that at x = 0, the synthesis potential of MgB2 has the most positive value, hence MgB2 will be the first (equilibrium) cathodic product. However, at a certain negative value of parameter x = x1 the line for the synthesis potential of MgB2 is crossed by the line for the deposition potential for Mg, and therefore at x < x1 Mg will be the equilibrium cathodic product. Parameter x1 can be calculated as:
On the other hand, at a certain positive value of parameter x = x2 the line of the synthesis potential for MgB2 is crossed by the line of the synthesis potential for MgB4 in Fig.1, and at x > x2 MgB4 will be the equilibrium cathodic product. The value of x2 can be calculated as:
Hence, the following conclusions can be made:
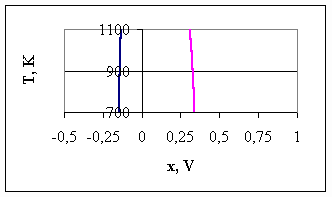
As discussed above, due to the lack of reliable data for thermodynamic properties of MgB6 and MgB12, values of x3, x4 and x5 cannot be determined in this paper. However, from the point of view of the range where MgB2 can be synthesised, this information is sufficient. The values of x1 and x2 are given in Table 3 and Fig.2, as function of temperature. One can see, that there is a very wide electrochemical window, from 442 mV (at 1,100 K) to 488 mV (at 700 K), within which MgB2 can be synthesised as a stoichiometric compound. Usually, an electrochemical window of about 100 mV is sufficient to conduct the synthesis in a stoichiometric regime. Therefore, the possibility of electrochemical synthesis of MgB2 layer from molten salts seems to be very promising. Using appropriate additives to the NaCl-KCl-MgCl2-KBF4 system, parameter x probably can be kept in the interval between the x1 and x2 values calculated in the present paper and thus, the synthesis of MgB2 will be possible. Table 3. The stability interval of MgB2 during electrochemical synthesis from molten salts
Fig.2. The Mg-rich side of the Mg-B EES diagram 4. Conclusions
References
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

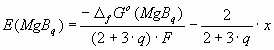 (1)
(1)
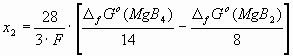 (3)
(3)