Tamás Fülöp, Tibor Egyházy, 1 István Sajó Department of Environmental Engineering and Chemical
Technology, 1 Bay Zoltán Institute of Materials Science and Technology, Budapest, HUNGARY
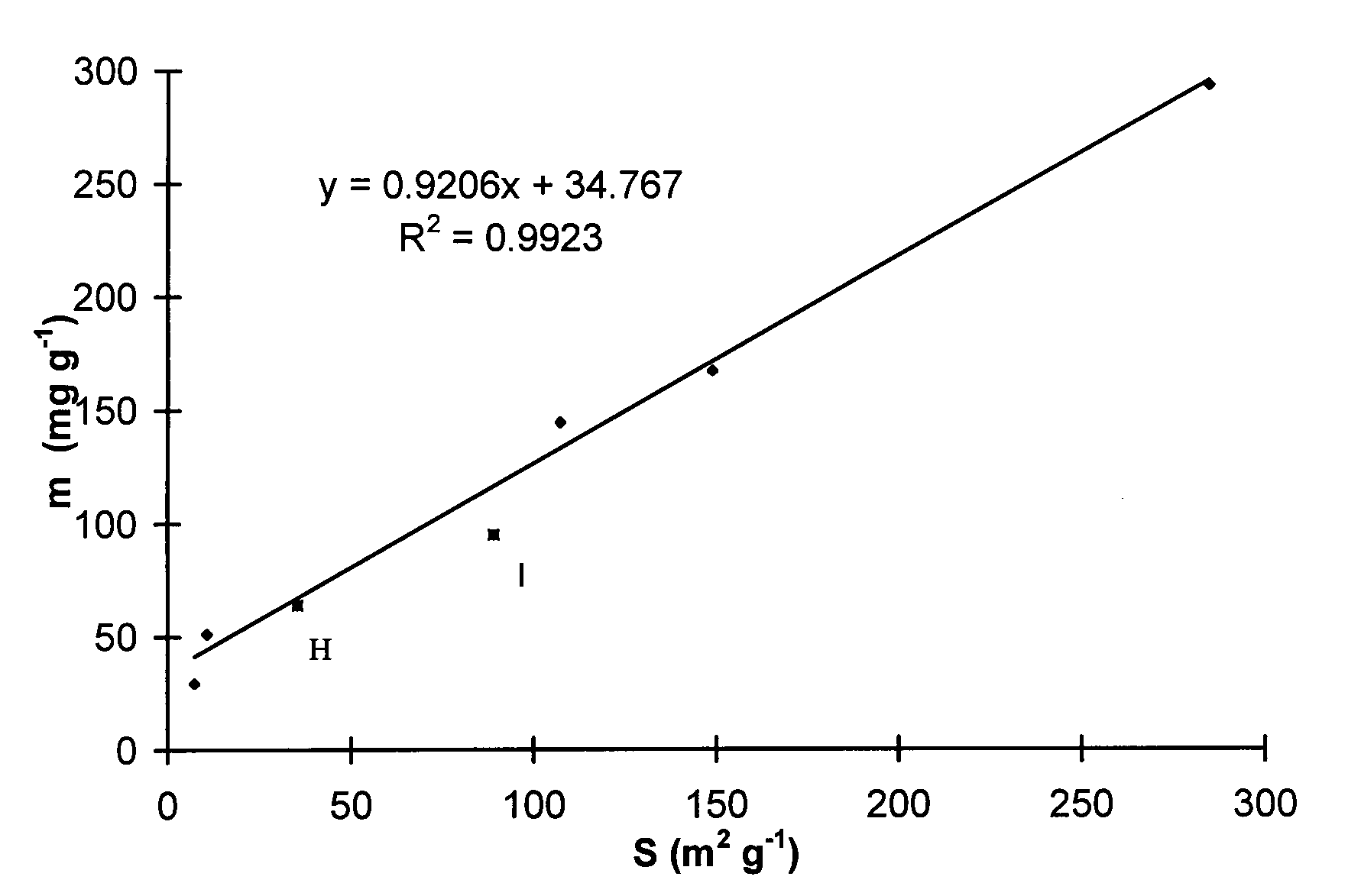
The study of alumina is of great importance because it is a large volume product on the chemical market. Although the major part of it is used in the production of aluminum metal, an increasing amount is employed in other fields, e. g. adsorbents, catalysts and catalyst carriers, ceramics, abrasives. Activated aluminas represent a group of technically important alumina chemicals. These products are obtained by thermal dehydration of different aluminum hydroxides in the 300-1000 °C temperature range. Water is driven out upon heating and a highly porous structure of aluminum oxide having a high surface area remains. Principal uses are as adsorbents, catalysts and catalyst carriers which are important applications in environmental protection. Alumina has several crystallographic modifications; these are designated by Greek letters. Among them g -alumina has the highest specific surface area. Above the calcination temperature of 1000°C the dominant form is corundum a -AI2O3 having low specific surface area value. In course of thermal dehydration, however, the so called transition or activated aluminas appear. In course of the experimental part of our work the hydrogen fluoride adsorption capacity of different aluminas was investigated as the function of the physico-chemical properties of the samples. The emission of HF is a great environmental problem in the primary aluminum industry, in aluminum smelters. The waste gas from the electrolysis cells is purified by contacting it with fresh smelter grade alumina usually in fluidised bed. The problem here is that the dominant phase in smelter grade alumina is corundum having low surface area so the adsorption process is not effective. That's why we compared the HF adsorption capacity of smelter grade and transition aluminas and tried to find correlations between the adsorption capacity and the physico-chemical properties of the samples. For the experiments smelter grade aluminas were supplied from Hungary (sample H) and Italy (sample I). Transition alumina sample supplied by Rhone Poulenc Company was used as well (RP). This latter sample was thermally treated at distinct temperatures for different periods of time. The specific surface area of the samples (S, m2/g) were determined by ASAP 2000 Micromeritics nitrogen adsorption porosimeter and X-ray diffraction images were prepared by Philips PW diffractometer. The phase composition of the samples were determined by the method of total profile fitting developed in ALUTERV Research Institute. This method is suitable for the determination of phase composition within 10 % deviation. HF adsorption experiments were carried out in a polyethylene reactor. 1 % HF in nitrogen was loaded to 0.25 g of alumina sample. The flow rate of the gas was 5 I/h. The residual HF content of the gas leaving the adsorbent was adsorbed in dilute NaOH solution and the concentration of fluoride ions in the solution was determined by photometric method. According to the results the adsorption breakthrough curves were drawn up and the adsorption capacity of the samples (m, mg/g) was calculated. Results are shown in Table 1 and Figure 1.
Table 1 Phase composition of alumina samples
Figure 1 Adsorption capacity as the function of specific surface area of the samples According to these results it can be stated that the transition alumina samples have higher capacity toward HF adsorption than smelter grade aluminas have. It is unambiguously proved by the results using smelter grade aluminas as well because the dominant phase in the Italian sample is alumina and it has higher capacity than the Hungarian type alumina. The adsorption efficiency in the aluminum smelter can be improved by introducing transition samples into the adsorption process as well. But the effect of the recycled spent transition alumina on the total electrolysis process must be investigated.
Copyright © 2000 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||